We can't find the internet
Attempting to reconnect
Something went wrong!
Hang in there while we get back on track

£165.81
CRC Press Current Topics in Nonclinical Drug Development: Volume 1
Price data last checked 71 day(s) ago - refreshing...
Price History & Forecast
Last 20 days • 20 data points (No recent data available)
Price Distribution
Price distribution over 20 days • 2 price levels
Price Analysis
Most common price: £166 (16 days, 80.0%)
Price range: £157 - £166
Price levels: 2 different prices over 20 days
Description
Product Specifications
- Brand
- CRC Press
- Format
- Hardcover
- ASIN
- 0367136279
- Domain
- Amazon UK
- Release Date
- 23 December 2020
- Listed Since
- 18 October 2019
Barcode
No barcode data available
Similar Products You Might Like
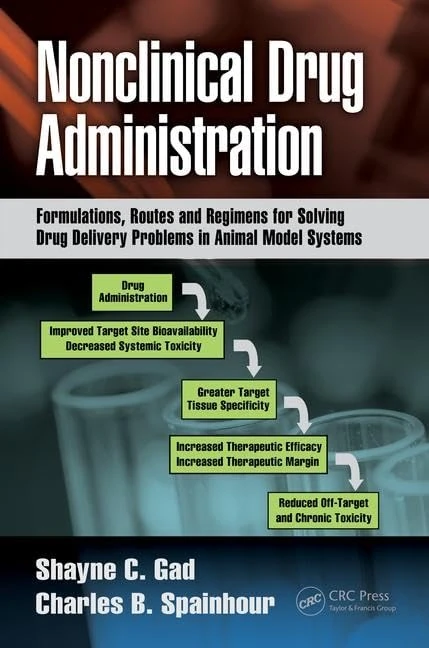
Nonclinical Drug Administration: Formulations, Routes and Regimens for Solving Drug Delivery Problems in Animal Model Systems
CRC Press

Contract Research and Development Organizations-Their History, Selection, and Utilization
Springer

Botanical Drug Products: Recent Developments and Market Trends
CRC Press

Recent Advancement in Prodrugs
CRC Press

Springer Nanophytomedicine: Concept to Clinic - Medical Book
Springer

Side Effects of Drugs Annual (Volume 41)
Elsevier

Development of Therapeutic Agents Handbook: 8 (Pharmaceutical Development Series)
Wiley

Annual Reports in Medicinal Chemistry (Volume 48)
Academic Press

Handbook of Research on Medicinal Chemistry: Innovations and Methodologies
CRC Press

Translating Molecules into Medicines: Cross-Functional Integration at the Drug Discovery-Development Interface: 25 (AAPS Advances in the Pharmaceutical Sciences Series, 25)
Springer

Side Effects of Drugs Annual (Volume 42)
Elsevier

Nanotoxicology: Toxicity Evaluation of Nanomedicine Applications

Multiregional Clinical Trials for Simultaneous Global New Drug Development (Chapman & Hall/CRC Biostatistics Series)
CRC Press

Immunotherapy – A Novel Facet of Modern Therapeutics
Springer

Immunotherapy – A Novel Facet of Modern Therapeutics
Springer

Pharmaceutical Dosage Forms - Parenteral Medications: Volume 3: Regulations, Validation and the Future
CRC Press

Springer - Nano-Biomaterials For Ophthalmic Drug Delivery Book
Springer

Recent Advances in Medicinal Chemistry, Volume 1
Elsevier

Nanobiotechnology: Human Health and the Environment
CRC Press

Bioactive Peptides: Production, Bioavailability, Health Potential, and Regulatory Issues (Nutraceuticals)
CRC Press

Fundamentals of Nanotoxicology: Concepts and Applications
Academic Press

CRC Press Natural Medicines: Clinical Efficacy, Safety and Quality
CRC Press

Academic Press Pharmacokinetics and Toxicokinetic Considerations II
Academic Press

Food Toxicology: Current Advances and Future Challenges
CRC Press