We can't find the internet
Attempting to reconnect
Something went wrong!
Hang in there while we get back on track

£80.77
Clinical Research Law and Ethics: Human Research and a Complete Practical Guide to Regulatory Compliance
Price data last checked 107 day(s) ago - refreshing...
We'll watch every seller, every day. One email when your price arrives.
This is the most expensive it has ever been. Walk away.
£81 today · previous high £81 · all-time low £77
NEW HERE?
Amazon shows you one price. We show you all of them.
Tosheroon watches Amazon prices so you don't have to. Every product on Amazon has a price history — we make it visible. Set the price you'd actually pay, and we'll email you the second it gets there. No app, no account, one email.
WHAT'S ON THIS PAGE
when this has been cheap or pricey
where the price is heading next
all-time high & low, recent range
name your number, we'll email you
Price History & Forecast
Grey patches = out of stock. Cheaper = lower on the chart. Hover for exact prices.
Last 567 days • 567 data points (No recent data available)
Price Distribution
Price distribution over 567 days • 2 price levels
Price Analysis
Most common price: £77 (426 days, 75.1%)
Price range: £77 - £81
Price levels: 2 different prices over 567 days
Description
Product Specifications
- Format
- paperback
- ASIN
- B0C12JG22P
- Domain
- Amazon UK
- Release Date
- 28 March 2023
- Listed Since
- 02 April 2023
Barcode
No barcode data available
Similar Products You Might Like

A Concise Guide to Clinical Trials
Wiley-Blackwell

A Clinical Trials Manual From The Duke Clinical Research Institute: Lessons from a Horse Named Jim
Wiley

Multiregional Clinical Trials for Simultaneous Global New Drug Development (Chapman & Hall/CRC Biostatistics Series)
CRC Press

Principles and Practice of Clinical Research
Academic Press

Essentials of Clinical Research
Springer

Clinical Trials Handbook: Design and Conduct
Wiley

A Practical Guide to Human Research and Clinical Trials
CRC Press

Clinical Trials and Tribulations
Academic Press

Clinical Trials of Drugs and Biopharmaceuticals
CRC Press

Publishing and Presenting Clinical Research
Lippincott Williams & Wilkins
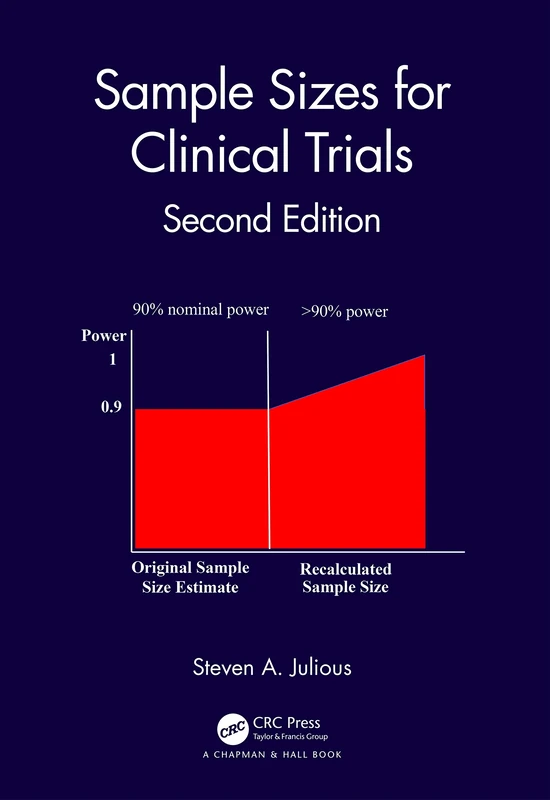
Sample Sizes for Clinical Trials
Chapman and Hall/CRC

Critical Thinking in Clinical Research: Applied Theory and Practice Using Case Studies
Oxford University Press

Principles and Practice of Clinical Research
Academic Press

Global Clinical Trials Playbook: Capacity and Capability Building
Academic Press

Principles and Practice of Clinical Trial Medicine
Academic Press

Design and Analysis of Clinical Trials: Concept and Methodologies (Wiley Series in Probability and Statistics)
Wiley-Blackwell

The Coordination of Clinical Research: A Handbook for Research Coordinators
Thieme

A Practical Guide to Managing Clinical Trials
CRC Press

Essentials of Clinical Research
Springer

Clinical Research Monitoring: A European Approach
World Scientific Publishing Company

Adaptive Design Theory and Implementation Using SAS and R (Chapman & Hall/CRC Biostatistics Series)
CRC Press

Adaptive Design Theory and Implementation Using SAS and R (Chapman & Hall/CRC Biostatistics Series)
CRC Press

Practical Guide to Clinical Data Management

Principles and Practice of Clinical Trials
Springer