We can't find the internet
Attempting to reconnect
Something went wrong!
Hang in there while we get back on track
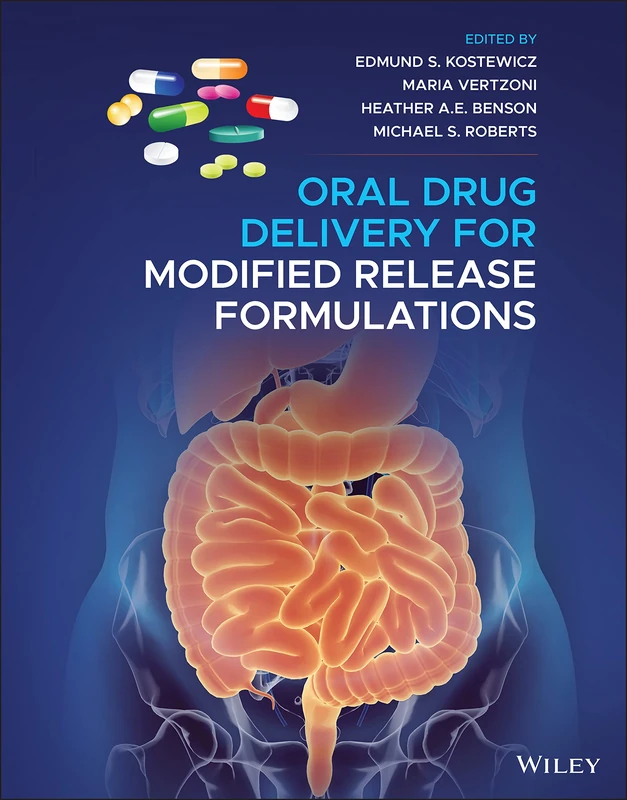
£138.00
Wiley Oral Drug Delivery for Modified Release Formulations
9781119772699
Price data last checked 135 day(s) ago - refreshing...
We'll watch every seller, every day. One email when your price arrives.
It has never been this cheap. We have no record of a lower price.
£138 today · cheaper than every other day in the last 23 months
NEW HERE?
Amazon shows you one price. We show you all of them.
Tosheroon watches Amazon prices so you don't have to. Every product on Amazon has a price history — we make it visible. Set the price you'd actually pay, and we'll email you the second it gets there. No app, no account, one email.
WHAT'S ON THIS PAGE
when this has been cheap or pricey
where the price is heading next
all-time high & low, recent range
name your number, we'll email you
Price History & Forecast
Grey patches = out of stock. Cheaper = lower on the chart. Hover for exact prices.
Last 543 days • 543 data points (No recent data available)
Price Distribution
Price distribution over 543 days • 8 price levels
Price Analysis
Most common price: £148 (277 days, 51.0%)
Price range: £138 - £153
Price levels: 8 different prices over 543 days
Description
Product Specifications
- Brand
- Wiley
- Model
- 9781119772699
- Format
- hardcover
- ASIN
- 1119772699
- Domain
- Amazon UK
- Release Date
- 15 April 2022
- Listed Since
- 13 November 2020
Barcode
No barcode data available
Similar Products You Might Like

Drug Delivery Approaches: Perspectives from Pharmacokinetics and Pharmacodynamics
Wiley

Modeling in Biopharmaceutics, Pharmacokinetics and Pharmacodynamics: Homogeneous and Heterogeneous Approaches: 30 (Interdisciplinary Applied Mathematics, 30)
Springer

McGraw-Hill Design of Controlled Release Drug Delivery Systems
McGraw-Hill Education

Biopharmaceutics Applications in Drug Development
Springer

Characterization of Pharmaceutical Nano- and Microsystems (Advances in Pharmaceutical Technology)
Wiley

Translational ADMET for Drug Therapy: Principles, Methods, and Pharmaceutical Applications
Wiley

Preformulation in Solid Dosage Form Development: 178 (Drugs and the Pharmaceutical Sciences)
CRC Press

Water-Insoluble Drug Formulation
CRC Press

Water-Insoluble Drug Formulation
CRC Press
Computational Pharmaceutical Solid State Chemistry
Wiley

Drug-Like Properties: Concepts, Structure Design and Methods from Adme to Toxicity Optimization
Academic Press

Developing Solid Oral Dosage Forms: Pharmaceutical Theory and Practice
Academic Press

Biopharmaceutics: From Fundamentals to Industrial Practice (Advances in Pharmaceutical Technology)

Shargel and Yu's Applied Biopharmaceutics & Pharmacokinetics, 8th Edition
McGraw-Hill Education

Polymeric Nanosystems: Theranostic Nanosystems, Volume 1
Academic Press

Nanotherapeutics in Cancer Vaccination and Challenges
Academic Press

Inorganic Nanosystems: Theranostic Nanosystems, Volume 2
Academic Press

Poorly Soluble Drugs: Dissolution and Drug Release (Pan Stanford Series on Pharmaceutical Analysis, 1)
Taylor & Francis

Assessing Bioavailablility of Drug Delivery Systems: Mathematical Modeling
CRC Press

Specification of Drug Substances and Products: Development and Validation of Analytical Methods
Elsevier

Oral Delivery of Therapeutic Peptides and Proteins
Academic Press

Physiological Pharmaceutics: Barriers to Drug Absorption
CRC Press

Immunotherapy – A Novel Facet of Modern Therapeutics
Springer

Essentials of Pharmaceutical Technology
MTG